To identify these double line structures, another quantum number is necessary. When spectral lines of hydrogen, lithium, sodium, and potassium are observed by the instrument of high resolving power, each of the lines of the spectral series was found to consist of a pair of lines known as a double line structure. Total number of m in a given l = (2l + 1) For a given value of l, m can have any integral value between +l to −l. The total values of m depend on the values of l. Magnetic quantum is denoted by the symbol ‘m’ or m l. This subdivision gives an additional quantum number which is called magnetic quantum numbers. Total number of subshells in the main shellīohr’s one electronic model could not explain the splitting of a single spectral line into a number of closely spaced lines in presence of a magnetic field or electric field.Īccording to Linde, the presence of more lines in the spectrum indicates that the energy levels are further subdivided. Various azimuthal quantum numbers and subshell designation is given below the table, Value of n and main shell designation 
Similarly, in the 2p designation, the number 2 stands for the value of n and the letter p denotes l = 1. In 1s designation, the number 1 stands for the value of n and the letter s denotes l = 0. Total values of l for a given value of n define the total number of subshells present in the main energy level of an atom. The total number of different values of l is equal to n. The azimuthal quantum number (l) can have any value from o to (n−1) for a given value of n. The azimuthal quantum number is denoted by the letter ‘l’. It describes the geometric shape of an orbital or electron wave. The azimuthal quantum numbers were introduced by Sommerfeld in his atomic structure to derive the angular momentum of an electron in an elliptical orbit. Therefore, if n = 3, the electron may reside in M-shell. When n = 1, 2, 3, …, the latter K, L, M, … are used to designate the value of n. The letter K, L, M, … are also used to derive the value of n. The principal quantum number (n) is always an integer having the value from 1 to ∞. Therefore, the size of the orbital also increases. As the value of n increases, the radius or nucleus-electron separation increases. For multielectron atoms, the energy of each electron depends mostly on the value of the principal quantum number (n).For the hydrogen atom, the energy is fixed because it contains only one electron.The principal quantum number is used to determine the size of an atom and the energy of an electron. The principal quantum number describes the set of energy levels or the principal shell to which an electron can stay. Total number of electrons in the main shell = 2n 2 Total number of electrons in s, p, d, f orbital = 2(2l + 1) Quantum numbers chart Principal quantum number (n) Therefore, we need four quantum numbers to explain various spectral lines in an atom of periodic table elements. 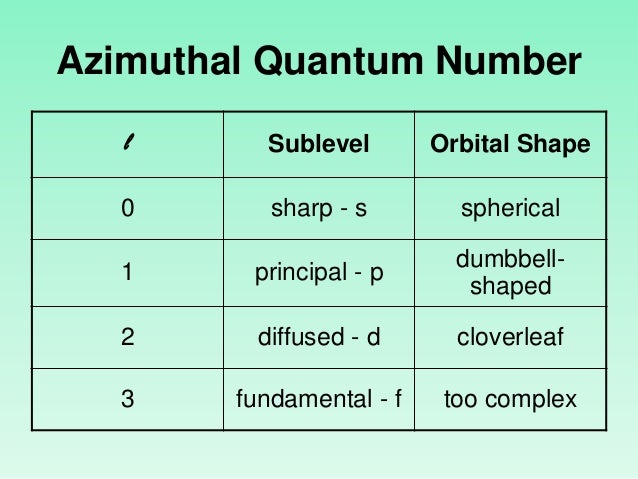
In learning chemistry or physics, it is not sufficient to explain the absorption or emission spectrum of various atoms. He indicates only one quantum number of an atom. We have seen that the Bohr model could not explain the fine structure of the hydrogen spectrum. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
